Overview
In this project, we had to design and create a board game with chemical reactions. My group made a game with the theme of Winter Wonderland. The end goal was to make it to the Christmas tree at the top, while doing chemical reactions along the way. We made 4 3D printed game pieces for our players to use, and they rolled dice to determine how far they would move. If they landed on a reaction square, we would guide them through the reaction. In these documents below, you can see our rules and guidelines for our game.
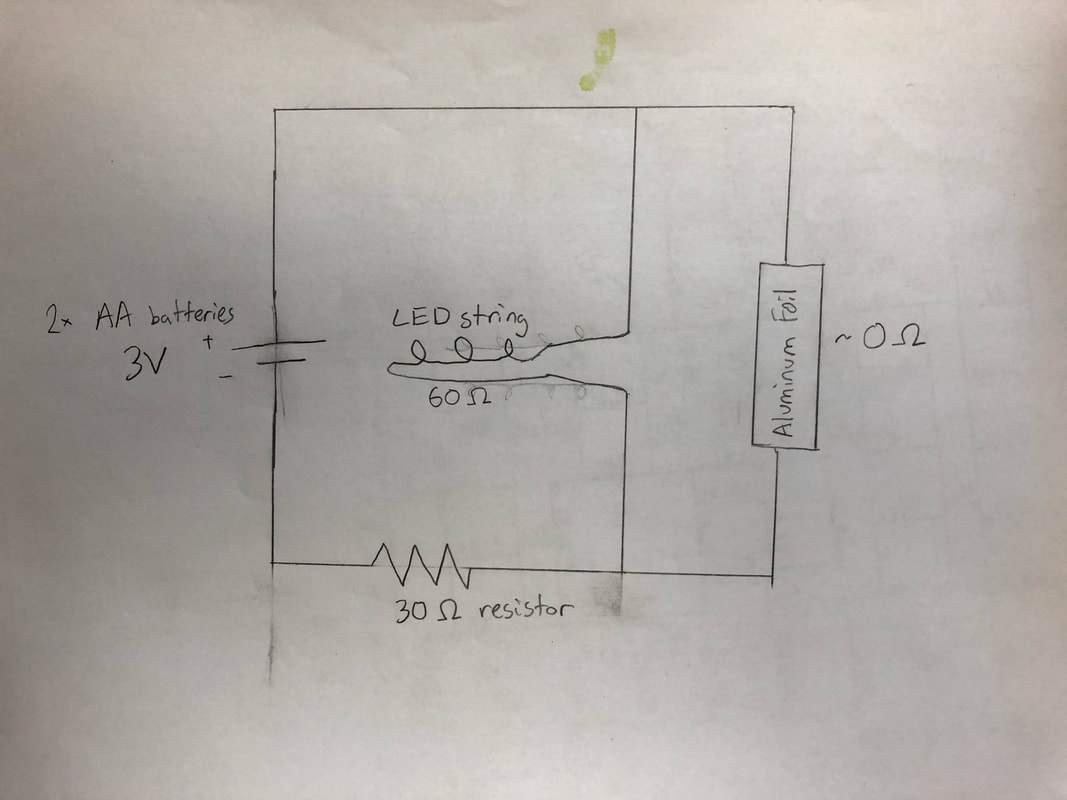
We wanted to make our game different so we came up with creative ideas, like 3D printing game pieces or using the lit LEDs to light up the tree.
Key Terms and Concepts
Ion - an atom that has lost or gained electrons (does not have a neutral charge)
Cation - a positively charged ion
Anion - a negatively charged ion
Compound - a substance formed when multiple chemical elements are chemically bonded together
Chemical reaction - a process that leads to the transformation of one set of chemical substances to another, creates a new substance. Ex. Burning, production of a gas
Physical reaction - a process that affects the form of a chemical substance, but not its chemical composition, no new substances are created. Ex. Cutting, ripping, boiling
Reactants - the things that interact with each other during a reaction and undergo a change
Products - the compounds or elements that are the result of a reaction
Single Replacement reaction - a type of chemical reaction where a cation reacts with a compound and takes the place of the cation in that compound
Double Replacement reaction - a type of chemical reaction where two compounds react, and the positive ions (cation) and the negative ions (anion) of the two reactants switch places, forming two new compounds or products
Decomposition reaction - a type of chemical reaction in which a single compound breaks down into smaller parts
Synthesis reaction - a type of reaction where multiple reactants combine and form a single product
Combustion reaction - a type of reaction when oxygen reacts with a "fuel" to create carbon dioxide and water
MSDS - material safety data sheet that outlines safety for chemicals
Cation - a positively charged ion
Anion - a negatively charged ion
Compound - a substance formed when multiple chemical elements are chemically bonded together
Chemical reaction - a process that leads to the transformation of one set of chemical substances to another, creates a new substance. Ex. Burning, production of a gas
Physical reaction - a process that affects the form of a chemical substance, but not its chemical composition, no new substances are created. Ex. Cutting, ripping, boiling
Reactants - the things that interact with each other during a reaction and undergo a change
Products - the compounds or elements that are the result of a reaction
Single Replacement reaction - a type of chemical reaction where a cation reacts with a compound and takes the place of the cation in that compound
Double Replacement reaction - a type of chemical reaction where two compounds react, and the positive ions (cation) and the negative ions (anion) of the two reactants switch places, forming two new compounds or products
Decomposition reaction - a type of chemical reaction in which a single compound breaks down into smaller parts
Synthesis reaction - a type of reaction where multiple reactants combine and form a single product
Combustion reaction - a type of reaction when oxygen reacts with a "fuel" to create carbon dioxide and water
MSDS - material safety data sheet that outlines safety for chemicals
Reflection
This project was super fun! My group and I worked well together and we came up with lots of creative ideas. One peak, or something that went well, in this project was our work ethic. We went in on Saturday Academy to work on our project more and we always did as much as we could during class. Additionally, we did some work at our houses too. The culmination of our efforts and hard work was evident in the presentation that we gave. I felt that we were very well prepared and we were able to add many extra things to our presentation, like 3D printed game pieces and a lit-up tree. One pit, or something that could've been better, was our time management. We definitely could have used more time near the end of our project to run through our game and test the rules. Once at game night, we realized that our game was too short to give players special boosts, so we removed it from our game. Another peak was our collaboration. We listened to everyone's ideas and because of this, were able to come up with the best possible ideas to use. No one's ideas got shut down. Lastly, another pit was finding our reactions. It was hard to decide which reactions we wanted to do, and we did not figure it out until the very last week. Relating this to the other pit, it probably would've been better if we had planned everything better. However, we did manage to find all the necessary reactions to do and our project worked.