Project Description
This project's goal was to create and design an outdoor classroom. We learned about atomic structure, state, and heat, how to design a solar water heater, and the solar angles of the sun. We also learned about site selection and how to design our classroom. Than we presented our designs.
Solar Water Heater
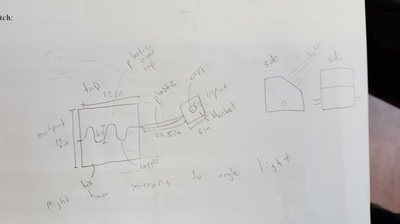
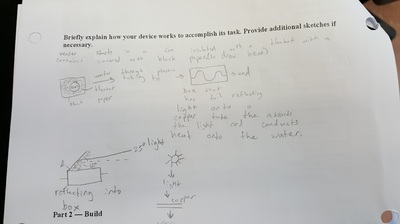
In preparation for designing our classroom, we designed and built a small solar water heater. We had to use the sun's rays to heat water and during this project, we used concepts from atomic structure and solar angles which you can find lower on this page. We decided to follow the guidelines we were given which consisted of a container that heats the water with the copper and a storage tank that feeds water to the container.
Design:
Here are the sketches and materials list we had. Basically, our water is poured into a can that is insulated and then it travels into a copper tube that goes through the box with reflective material that was designed to reflect light onto the pipe. At the end we circulated the water back to the begining.
Here are the sketches and materials list we had. Basically, our water is poured into a can that is insulated and then it travels into a copper tube that goes through the box with reflective material that was designed to reflect light onto the pipe. At the end we circulated the water back to the begining.
End Result: We got the water temperature to rise by 5 degrees Celsius for 100 mL of water. We calculated that this would be an energy gain of 2,093 Joules. Our group's design wasn't the most efficient but it wasn't the least. The most efficient ones were the ones who kept water in their heater the whole time, without the error of leaking and human error.
Site Selection
We picked four places on campus and rated them based on the qualities we thought were important. We rated wind, wi-fi, sunlight, shade, accessibility, visibility, ground cover, and view. In the end, we decided that the foundation between the cafeteria and the arts building because we decided it had close proximity to wi-fi, low wind, good sunlight, and it was usable and accessible by the students and staff. This also satisfied our client needs portion, which you can read about below.
Material Testing
We tested numerous materials for heat conductivity and water absorption. We ended up using pressure treated wood for our wood because of the outdoor conditions it would have to face. We also decided that wood chips could be a suitable ground cover.
Design with Client Needs
To start our design, we first conducted a client needs survey and we found what our clients want. The survey was at tinyurl.com/stemoutdoor.
110 people took the survey.
We found that people want:
110 people took the survey.
We found that people want:
- Wi-fi(75 ppl)
- Whiteboard(38 ppl)
- Group Work(67 ppl)
- Shaded Area(62 ppl)
Presentation
Model Shade Structure
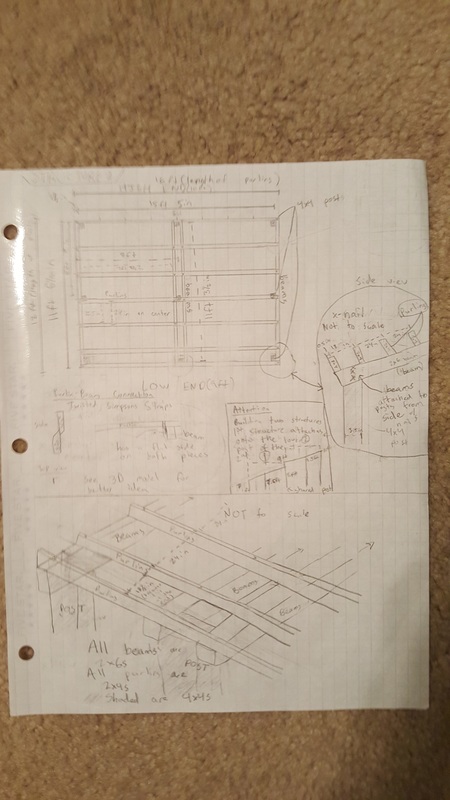
Here is a picture of the model shade structure to scale. The paper bag layer is the ground and you can see how far into the ground the posts go.
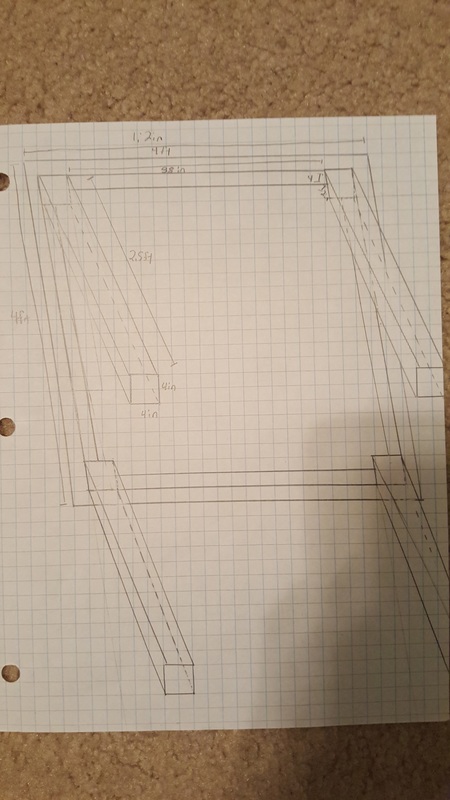
Blueprints
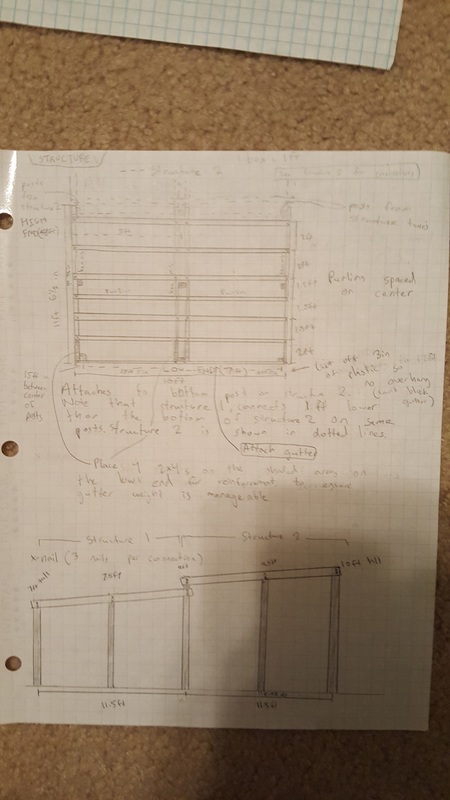
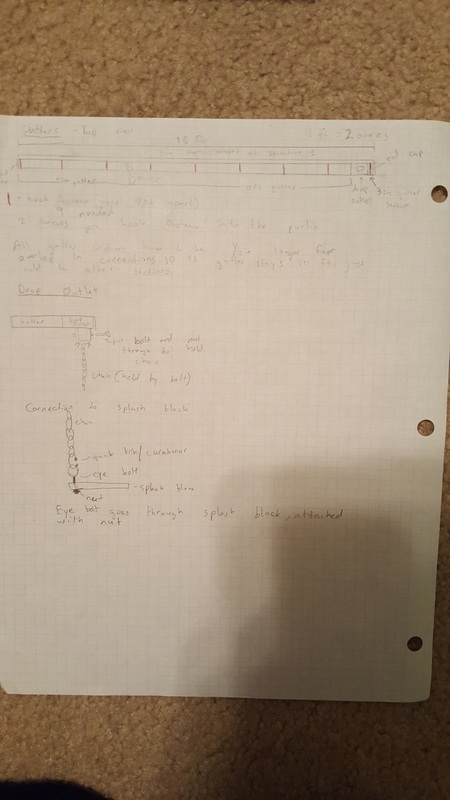
We made blueprints for the tables, cabinets, and shade structure.
Wind Turbine Lab
We did a lab about wind turbines to try to figure out the most efficient design for VAWT(vertical axis wind turbine) and HAWT(horizontal axis wind turbine). We measured efficiency on voltage. Most groups figured out that aluminium was the best material and that pinwheels are the best design, especially at high wind speeds and on HAWT. The reason why was because aluminum is rigid, so no energy is lost in the material bending. Pinwheels worked the best because they can spin the fastest because of their wind pockets and air flow. The reason why we don't see giant pinwheels on wind farms is because generating energy isn't actually based on voltage or RPM, it is based on speed, and the three bladed pinwheels have a very high speed on the outer tip of each blade.
Energy Justification
Here is a link to the energy justification document our class made to justify why we should use renewable energy and make an outdoor classroom. https://docs.google.com/document/d/1bFY3_1XyQnYaHe64XkJ7PyTiK26ZoMtmcsY96jM8fSQ/edit
Concepts
Solar Angles
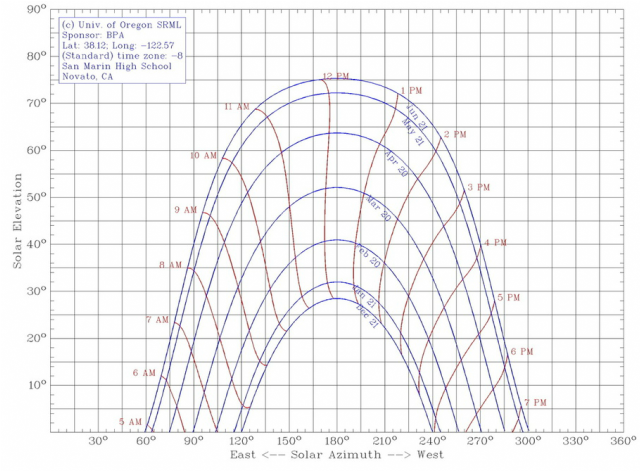
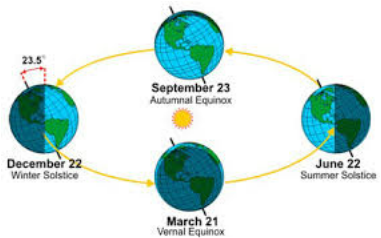
The Sun's rays fall differently onto the Earth each day because of the rotations and revolutions of the Earth and Sun along with the Earth being tilted at 23.5 degrees. As seen in the diagram on the right, the northern hemisphere will have more direct light in summer and less in winter. The effect of this is shown in the diagram on the left. In June, the sun is higher in the sky, while it is lower in January.
Atomic Structure
Atoms are the basic structure/building block of all matter.
-protons- have a positive charge, in nucleus
-neutrons- neutral charge, in nucleus
-electrons- no mass, negative charge, orbits nucleus
The number of protons is the atomic number. The number of protons and neutrons combined is the atomic mass.
Isotope: an atom with an unusual number of neutrons
Ion: an atom with an unusual number of electrons
Atoms usually have an equal number of protons, neutrons, and electrons if they aren't an ion or an isotope.
Molecule: 2 or more atoms bonded together
Compound: a molecule made of different elements
States of matter:
solids: Molecules are bonded firmly and hold densely. They hold the least energy/heat.(ex. ice)
liquids: Molecules are bonded and can move around and change bonds. Holds more energy/heat.(ex. water)
gases: Molecules are very rarely bonded(1/100th as dense as solids). Most energy/heat.(ex. steam)
-protons- have a positive charge, in nucleus
-neutrons- neutral charge, in nucleus
-electrons- no mass, negative charge, orbits nucleus
The number of protons is the atomic number. The number of protons and neutrons combined is the atomic mass.
Isotope: an atom with an unusual number of neutrons
Ion: an atom with an unusual number of electrons
Atoms usually have an equal number of protons, neutrons, and electrons if they aren't an ion or an isotope.
Molecule: 2 or more atoms bonded together
Compound: a molecule made of different elements
States of matter:
solids: Molecules are bonded firmly and hold densely. They hold the least energy/heat.(ex. ice)
liquids: Molecules are bonded and can move around and change bonds. Holds more energy/heat.(ex. water)
gases: Molecules are very rarely bonded(1/100th as dense as solids). Most energy/heat.(ex. steam)
Heat
Specific heat capacity:
The specific heat is the amount of heat per unit mass required to raise the temperature by one degree Celsius. A higher specific heat means that it is harder to heat a material.
Heat transfer:
Conduction: heat transfer through solids
Convection: heat transfer through a fluid(liquid/gas)
Radiation: heat transfer through waves or ray(no medium required)
Insulation is the opposite of conduction.
Conduction: good at transferring heat.
Laws of Thermodynamics:
0th law: If two systems are in thermal equilibrium with a third, then they are in equilibrium with each other. (A=B and B=C, so A=C)
1st law: Conservation of Energy: energy is neither created or destroyed. but transferred from one form to another. Heat is a form of energy.
2nd law: Entropy increases. Over time, everything becomes the same temperature.
3rd law: Temperature can never reach absolute zero. Heat always exists.
The specific heat is the amount of heat per unit mass required to raise the temperature by one degree Celsius. A higher specific heat means that it is harder to heat a material.
Heat transfer:
Conduction: heat transfer through solids
Convection: heat transfer through a fluid(liquid/gas)
Radiation: heat transfer through waves or ray(no medium required)
Insulation is the opposite of conduction.
Conduction: good at transferring heat.
Laws of Thermodynamics:
0th law: If two systems are in thermal equilibrium with a third, then they are in equilibrium with each other. (A=B and B=C, so A=C)
1st law: Conservation of Energy: energy is neither created or destroyed. but transferred from one form to another. Heat is a form of energy.
2nd law: Entropy increases. Over time, everything becomes the same temperature.
3rd law: Temperature can never reach absolute zero. Heat always exists.
Reflection
Overall, this was a very good project. I enjoyed many parts of it and I learned a lot of concepts and information about design and building. One peak, or something good, was that I learned a lot about building design and how to construct wooden structures. My job in our group was to design the shade structure and I started off with a general idea of a slanted roof with corrugated plastic over it. I soon learned that this would be a lot harder to design, because I had to make it stand above the ground, requiring many posts. This also required me to search span calculators and make sure that the beams weren't spanning too far. Connecting all of the pieces together also required additional research. I had to use Simpsons straps to connect the beams and purlins, as you can see in our presentation. I had never seen a Simpsons strap before and now I see how useful they are. One pit, something that could have gone better, was work productivity. We had trouble getting everyone in our group to work, so we fell behind schedule and ended up working to the very last day right before the presentation. One peak out of this was that we were able to focus when we really needed to and we were able to gain back all of the lost unproductive hours. I think it was a good learning experience for us to see how we could manage our time better. The last pit was our presentation structure and content. I feel that we could have improved on our presentation and structured it better if we had more productive hours to run through it multiple times. We had a set 5 minute time cap and our presentation had too much info, making us have to talk too quickly which lowered our communication scores. We should have spent more time on refining the presentation and only focusing on the key points. In conclusion, this project was very successful and we learned a lot from new experiences and the challenges we had to overcome. We also learned what we could have improved on in order to make our presentation better. We can use this info in the future.